The Architecture of Orthodontic Lab Quality Control

Predictability in orthodontic outcomes hinges on a systematic quality assurance framework that validates digital data, manufacturing parameters, and biocompatibility long before an appliance reaches your clinic. While the transition to a digital workflow from intraoral scans has significantly reduced manual error, it has introduced sophisticated complexities in data integrity and material science. Modern lab quality control is no longer a simple visual check of a stone model; it is a multi-stage validation process essential for maintaining precision in the modern practice.
The Foundation of Lab Compliance
In the European regulatory landscape, orthodontic appliances are classified as medical devices, making high manufacturing standards a regulatory necessity rather than an elective choice. Professional laboratories in the Baltics and Scandinavia typically align their operations with ISO 13485:2016. This international standard provides the necessary framework for traceability and process validation, ensuring that every device can be linked back to specific raw material batches and specific manufacturing conditions.
Traceability is particularly vital when utilizing specialized orthodontic 3D printing materials. The chemical integrity and mechanical properties of the final product depend entirely on precise curing cycles and controlled storage environments. Without rigorous documentation of these variables, the risk of material failure or leaching increases, potentially compromising patient safety and treatment efficacy.
A Systematic Three-Stage Inspection Workflow
A robust quality control system operates through three distinct phases: pre-production, in-process, and post-production. Each phase serves as a gatekeeper to ensure that the final appliance matches your clinical intent.
Digital Data Integrity and Pre-Production
The accuracy of the final appliance is dictated by the quality of the initial data. The first quality control gate occurs at the moment of file receipt, where technicians perform a comprehensive mesh analysis. This involves checking STL or PLY files for holes, noise, or "double walls" that could compromise the printing process.
Beyond technical file integrity, technicians perform an anatomical verification. They ensure the intraoral scan has accurately captured critical landmarks, such as terminal molars and the gingival zenith, which are essential for appliance retention. Finally, a senior technician reviews the CAD design to ensure the staging speeds for tooth movement are biologically sound and align with your specific prescription.
Manufacturing Validation and In-Process Monitoring
As labs move toward the 3D printing revolution in orthodontics, the focus shifts from manual craftsmanship to process validation. This requires proving that the manufacturing method consistently produces the intended result through regular equipment calibration.
Printers undergo frequent checks of light intensity and XY resolution to ensure that a 0.5mm movement in the planning software translates to exactly 0.5mm in the physical device. Furthermore, environmental controls are monitored strictly; fluctuations in temperature and humidity can alter resin viscosity, leading to dimensional drift that might not be visible to the naked eye but can cause significant fit issues chairside.

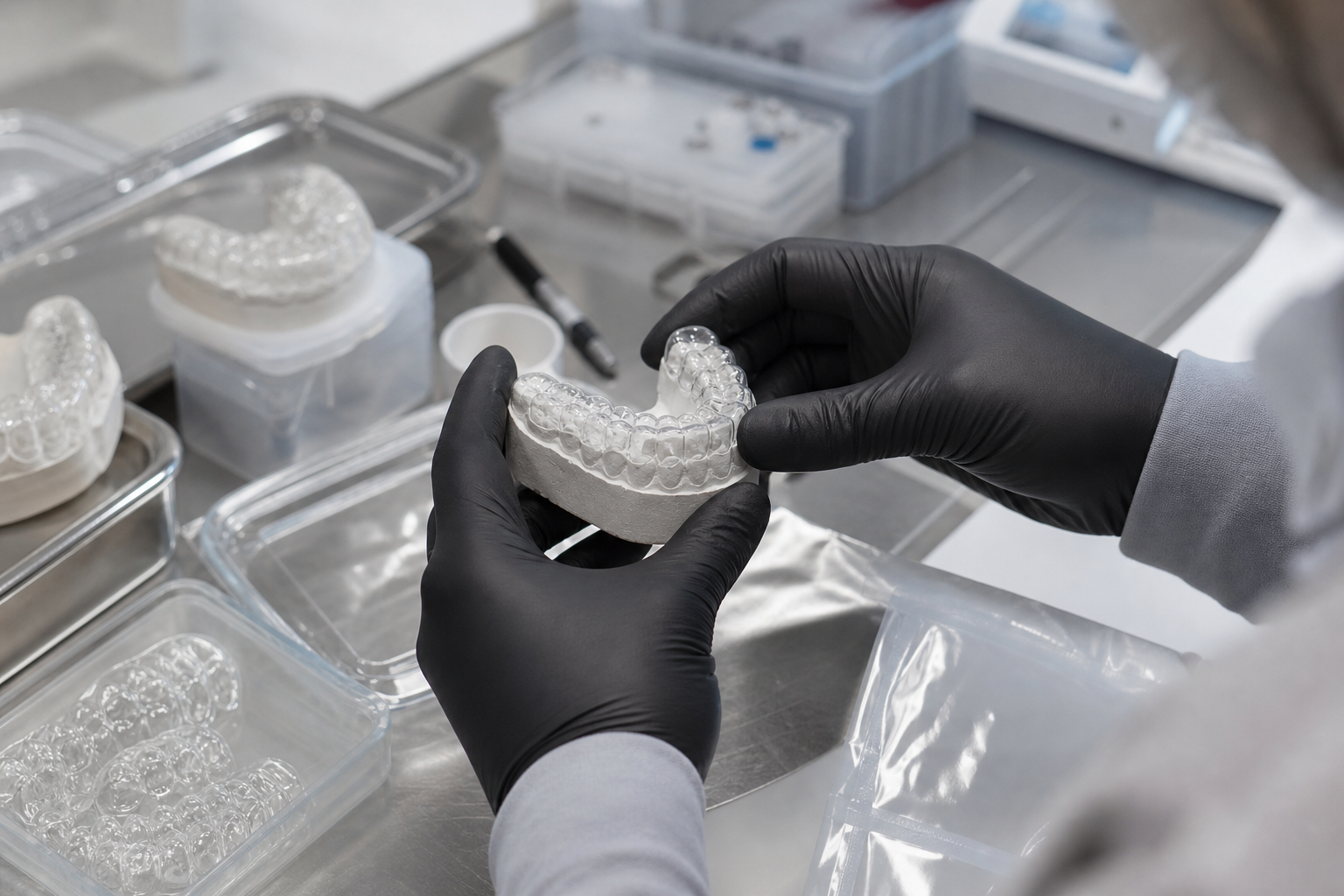
Physical Inspection and Post-Production Finishing
Even the most advanced additive manufacturing requires meticulous post-processing. This final stage is where many common quality issues, such as delamination or improper polymerization, are prevented. Quality control protocols verify that appliances spend the exact required duration in nitrogen-purged curing chambers to ensure full biocompatibility.
While physical models are used less frequently today, many labs maintain a "master model" strategy for complex appliances to verify the path of insertion and clasp tension. Before a device is sealed in medical-grade packaging, it undergoes a final visual inspection for surface finish, clarity, and edge smoothness to ensure maximum patient comfort.
Addressing the Automation Objection
A common concern among clinicians is that increased automation might replace the critical "human eye." It is tempting to assume that because a machine prints a file, the output is inherently perfect. However, digital systems can fail in subtle ways; software glitches can miscalculate wall thickness, or a dirty optical window can cause scaling errors.
The most effective quality systems use automation to handle high-volume production while reserving human expertise for strategic validation points. For example, the adoption of direct 3D-printed orthodontic appliances removes the manual variability associated with traditional thermoforming. While this eliminates many analog errors, it simultaneously increases the need for stringent chemical and mechanical testing to ensure the printed resin performs predictably in the oral environment.
Why Quality Control Impacts Clinic Profitability
For the modern clinician, a laboratory’s quality control protocol serves as an insurance policy against chairside failure. Every remake represents more than just a lost lab fee; it consumes valuable chair time, erodes patient trust, and disrupts practice momentum. Precision is the primary driver of efficiency in complex cases.
The evolution of clear aligners demonstrates that the difference between a successful treatment and a mid-course correction often lies in the precision of the manufacturing stage. When evaluating a lab partner, transparency regarding their staging algorithms and material certifications is essential. By choosing a partner committed to rigorous validation, you ensure that your clinical expertise is supported by high-accuracy, digitally-validated appliances.
To see how these quality standards are applied to modern appliance manufacturing, explore our integrated additive manufacturing solutions for orthodontics.
NordicDens is a modern orthodontic laboratory in Tallinn, Estonia, serving clinics across the Nordics and Europe with precision appliances and digital workflows.


